COVID-19 Alaska Clinical Update – July 28Thursday, July 28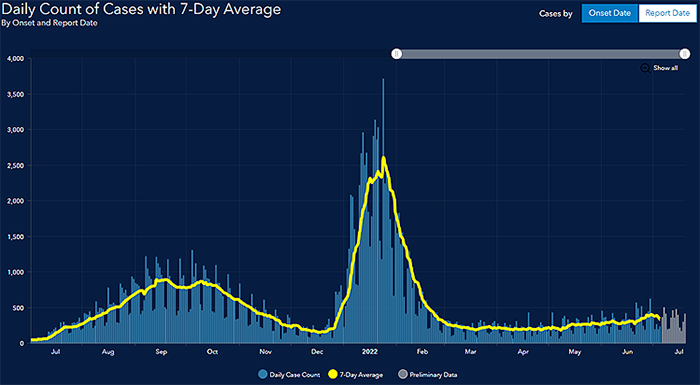
Graph taken from State of Alaska’s COVID-19 Cases Dashboard
Articles summarized in this update are compiled from a variety of sources but draw heavily on resources highlighted in the Johns Hopkins Center for Health Security’s weekly COVID-19 update.
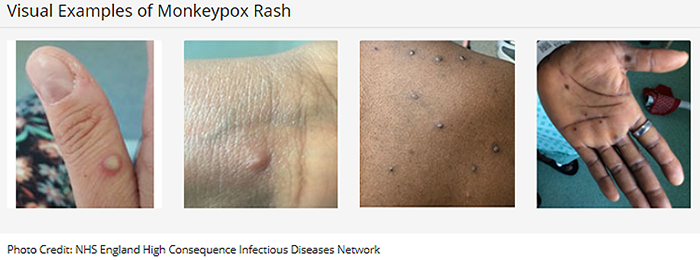
FDA authorizes Moderna and Pfizer-BioNTech COVID-19 vaccines for children 6 months of age and olderOn June 18 the CDC and FDA authorized emergency use of the Moderna and Pfizer-BioNTech COVID mRNA vaccines for children down to 6 months of age. For the Moderna COVID-19 Vaccine, the FDA amended the emergency use authorization (EUA) to include use of the vaccine in individuals 6 months through 17 years of age. The vaccine had been authorized for use in adults 18 years of age and older. The Moderna COVID-19 Vaccine is administered as a primary series of two doses, one month apart. For the Pfizer-BioNTech COVID-19 Vaccine, the FDA amended the EUA to include use of the vaccine in individuals 6 months through 4 years of age. The vaccine had been authorized for use in individuals 5 years of age and older. The Pfizer-BioNTech COVID-19 Vaccine is administered as a primary series of three doses in which the initial two doses are administered three weeks apart followed by a third dose administered at least eight weeks after the second dose. All children, including children who have already had COVID-19, should get vaccinated. On July 8, the FDA fully approved the Pfizer-BioNTech COVID-19 mRNA vaccine for individuals age 12-15 years. The vaccine was previously made available to this age group under emergency use authorization. FDA authorizes pharmacists to prescribe Paxlovid with certain limitationsOn July 6, the FDA revised the Emergency Use Authorization for Paxlovid, a COVID antiviral combination medication, to allow state-licensed pharmacists to prescribe Paxlovid to eligible patients. The authorization limits pharmacists to prescribe the medication if they are able to sufficiently assess renal and hepatic function as well as drug-drug interactions (i.e., through consultation with the patient’s established health care provider or access to recent health records). Pharmacists must refer patients for clinical evaluation if sufficient information is not available or if medication adjustments are needed due to a potential drug-drug interaction. Novavax COVID-19 vaccine authorizedOn July 19, the CDC authorized the Novavax COVID-19 vaccine as another primary series option for adults ages 18 years and older. The Novavax vaccine is the fourth COVID vaccine available in the USA. It contains nanoparticles made up of proteins from the surface of the coronavirus, a fundamentally different design than the three shots currently available in the United States, and a technology that is more traditional for vaccine development than the mRNA vaccines. This is the first COVID-19 protein subunit vaccine recommended for use in the United States that is built on a familiar type of vaccine technology. Other protein subunit vaccines have been used for more than 30 years in the United States, including for hepatitis B, HPV, and acellular pertussis (whooping cough) vaccines. WHO declares monkeypox a Public Health Emergency of International ConcernOn July 23, the WHO’s Director General declared monkeypox a Public Health Emergency of International Concern (PHEIC), which is the “loudest alarm bell the organization can ring.” Worldwide there have been more than 15,000 cases of monkeypox so far in more than 70 countries. Preparing for monkeypox in AlaskaNo cases of monkeypox have been detected in Alaska as of July 12, 2022. The Division of Public Health has published an information page on monkeypox with education for healthcare professionals and FAQs. In addition, an Alaska Public Health Alert was released on July 13 providing an update on testing and vaccination for monkeypox. Health care providers in Alaska should report suspected monkeypox cases to SOE immediately; please call SOE at 907-269-8000, or 1-800-478-0084 after hours. SOE can assist providers in obtaining appropriate clinical specimens that should be routed to the Alaska State Public Health Laboratory (ASPHL) in Anchorage for testing. Post-exposure vaccination should be prioritized for close contacts to cases. As supplies allow, vaccine might also be available for persons with certain high-risk exposures, such those who have had multiple sexual partners during the past 14 days in a community where monkeypox transmission is occurring or while attending an event associated with a monkeypox outbreak. - The rash associated with monkeypox involves vesicles or pustules that are deep-seated, firm or hard, and well-circumscribed; the lesions may umbilicate or become confluent and progress over time to scabs.
- Presenting symptoms typically include fever, chills, the distinctive rash, or new lymphadenopathy; however, onset of perianal or genital lesions in the absence of subjective fever has been reported.
- The rash associated with monkeypox can be confused with other diseases that are encountered in clinical practice (e.g., secondary syphilis, herpes, chancroid, and varicella zoster). However, a high index of suspicion for monkeypox is warranted when evaluating people with a characteristic rash, particularly for men who report sexual contact with other men and who present with lesions in the genital/perianal area or for individuals reporting a significant travel history in the month before illness onset or contact with a suspected or confirmed case of monkeypox.
Additional information and pictures can be found on the CDC website for Monkeypox Signs and Symptoms
 Omicron BA.5 sub variant accounts for more than 81% of COVID cases in USDriven by the Omicron subvariant BA.5, COVID cases and hospitalizations nationwide are at their highest levels since February 2022. For the week July 17-23, the BA.5 subvariant accounted for an estimated 81.9% of new SARS-CoV-2 infections. According to CDC Director Dr. Rochelle Walensky, "We do not know about the clinical severity of BA.4 and BA.5 in comparison to our other Omicron subvariants…But we do know it to be more transmissible and more immune-evading. People with prior infection, even with BA.1 and BA.2, are likely still at risk for BA.4 or BA.5." Because most COVID testing is now occurring with rapid at-home antigen tests that are never reported to public health departments, it is estimated that for every reported case there are 7-10 unreported cases. COVID-19 was third leading cause of death in the US in 2020 and 2021In 2020, heart disease and cancer were the leading causes of death in the US, accounting for 1.29 million deaths, followed by COVID-19, accounting for 350,000 deaths. From March 2020 to October 2021, heart disease (20.1%), cancer (17.5%), COVID-19 (12.2%), accidents (6.2%), and stroke (4.7%) were the most common causes of death in the US. There were 2.875 million deaths in March to December 2020 and 2.855 million deaths in January to October 2021; the 5 leading causes of death were the same in each year. From March 2020 to October 2021, COVID-19 accounted for 1 in 8 deaths in the US. Compared with the 2020 time period, deaths from COVID-19 in the 2021 time period decreased in ranking among those aged 85 years or older but increased in ranking among those aged 15 to 54 years, and became the leading cause of death among those aged 45 to 54 years. COVID-19 vaccines estimated to have prevented nearly 20 million deathsUsing a mathematical model of COVID-19 transmission and vaccination fit to reported COVID-19 mortality and all-cause excess mortality in 185 countries, authors estimated the number of deaths prevented by COVID-19 vaccination. The researchers estimated that vaccinations prevented 14.4 million (95% credible interval [Crl] 13.7–15.9) deaths from COVID-19 in 185 countries and territories between Dec 8, 2020, and Dec 8, 2021. This estimate rose to 19.8 million (95% Crl 19.1–20.4) deaths from COVID-19 averted when we used excess deaths as an estimate of the true extent of the pandemic, representing a global reduction of 63% in total deaths (19.8 million of 31.4 million) during the first year of COVID-19 vaccination. In low-income countries, we estimated that an additional 45% (95% CrI 42–49) of deaths could have been averted had the 20% vaccination coverage target set by COVAX been met by each country, and that an additional 111% (105–118) of deaths could have been averted had the 40% target set by WHO been met by each country by the end of 2021. More lives could have been saved if vaccines had been distributed more rapidly to many parts of the world and if vaccine uptake could have been strengthened worldwide. Limited availability for monoclonal antibody therapiesHHS Administration for Strategic Preparedness and Response (ASPR) announced that its supply of bebtelovimab, Eli Lilly’s the monoclonal antibody therapy, is expected to be depleted by the end of August 2022. ASPR is currently distributing about 30,000 doses per week from the federal government’s purchase of 750,000 doses. Eli Lilly is expected to make bebtelovimab commercially available for purchase through a distributor by the middle of August to ensure continuous availability of the therapy after the federal government’s purchased supply is depleted. Global real-world vaccine effectiveness against SARS-CoV-2 is 91-93% protecting against hospitalization and death from COVID-19 regardless of ageIn an expert review of 79 studies on vaccine effectiveness (VE) after primary immunization, the most-studied COVID-19 vaccines provide consistently high protection against serious clinical outcomes like hospitalizations and deaths, regardless of variant. VE was high and comparable for both AstraZeneca and mRNA vaccine types (91–93%) in protecting against hospitalization and death from COVID-19, regardless of age. VE against symptomatic infections trended higher (though not significantly) for mRNA-based vaccines compared to AstraZeneca. Waning of VE since time of vaccination was observed for symptomatic infections but was limited for serious COVID-19 outcomes. Maternal vaccination ranged from 38-80% effective at preventing COVID-19 hospitalization for infants younger than 6 monthsIn this case-control test-negative study, authors matched infants hospitalized with COVID-19 and infants hospitalized without COVID-19 to assess the effectiveness of maternal vaccination during pregnancy against hospitalization. A total of 537 case infants (181 of whom had been admitted to a hospital during the delta period and 356 during the omicron period; median age, 2 months) and 512 control infants were enrolled and included in the analyses; 16% of the case infants and 29% of the control infants had been born to mothers who had been fully vaccinated against Covid-19 during pregnancy. Two case infants died from Covid-19; neither infant’s mother had been vaccinated during pregnancy. The effectiveness of maternal vaccination against hospitalization for Covid-19 among infants was 52% (95% confidence interval [CI], 33 to 65) overall, 80% (95% CI, 60 to 90) during the delta period, and 38% (95% CI, 8 to 58) during the omicron period. Effectiveness was 69% (95% CI, 50 to 80) when maternal vaccination occurred after 20 weeks of pregnancy and 38% (95% CI, 3 to 60) during the first 20 weeks of pregnancy. The finding that the risk of hospitalization, and in particular, hospitalization in an ICU, was reduced among infants whose mothers had been fully vaccinated during pregnancy provides evidence of additional benefits of maternal vaccination beyond those previously reported for the mother. Prior vaccination, previous infection, and hybrid immunity all showed >70% effectiveness against severe, critical, or fatal COVID-19 due to omicron infectionIn this study, authors from Qatar conducted a national, matched, test-negative, case–control study in Qatar from December 23, 2021, through February 21, 2022, to evaluate the effectiveness of vaccination with Pfizer–BioNTech or Moderna COVID mRNA vaccines, natural immunity due to previous infection with variants other than omicron, and hybrid immunity (previous infection and vaccination) against symptomatic omicron infection and against severe, critical, or fatal COVID-19. The effectiveness of previous infection alone against symptomatic omicron (subvariant BA.2) infection was 46.1% (95% confidence interval [CI], 39.5 to 51.9). The effectiveness of vaccination with two doses of Pfizer-BioNTech and no previous infection was negligible (−1.1%; 95% CI, −7.1 to 4.6), but nearly all persons had received their second dose more than 6 months earlier. The effectiveness of three doses of Pfizer-BioNTech and no previous infection was 52.2% (95% CI, 48.1 to 55.9). The effectiveness of previous infection and two doses of Pfizer-BioNTech was 55.1% (95% CI, 50.9 to 58.9), and the effectiveness of previous infection and three doses of Pfizer-BioNTech was 77.3% (95% CI, 72.4 to 81.4). No notable differences were observed between the effects of Pfizer-BioNTech and Moderna vaccination. The protection conferred by hybrid immunity of previous infection and two-dose vaccination was similar to that of previous infection alone, at approximately 50%, which suggests that this protection originated from the previous infection and not from vaccination. No discernable differences in protection against symptomatic BA.1 and BA.2 infection were seen with previous infection, vaccination, and hybrid immunity. Vaccination enhanced protection among persons who had had a previous infection. Hybrid immunity resulting from previous infection and recent booster vaccination conferred the strongest protection. The highest effectiveness was seen with hybrid immunity from previous infection and recent booster vaccination (approximately 80%). This finding provides evidence for the benefit of vaccination, even for persons with a previous infection. Low rate of COVID rebound symptoms following Paxlovid treatmentIn this retrospective cohort study from the Mayo Clinic, authors found that only 4 of 483 high-risk patients (0.8%) experienced rebound of symptoms after being treated with Paxlovid. Rebound symptoms were generally mild, occurred at a median of 9 days after treatment, and all patients recovered without additional treatment. The study took place from February-April 2022. 93% of the patients in the cohort had been vaccinated, and many had received their booster doses. The median age was 63. All patients were considered at high-risk for severe disease (an EUA eligibility requirement), and none were immunocompromised. Two patients in the cohort were hospitalized, but for reasons other than COVID-19. All patients recovered from their COVID-19 infections. MMWRs<1% of Paxlovid-treated patients had COVID-related hospital admissions or ED encounters 5-15 days after treatmentIn this MMWR, authors used data from Kaiser Permanent Southern California to describe hospital admissions and emergency department (ED) encounters related to SARS-CoV-2 infections during the 5–15 days after pharmacy dispensation of a 5-day treatment course of Paxlovid. Among 5,287 persons aged ≥12 years who received Paxlovid during December 31, 2021–May 26, 2022, 73% had received ≥3 doses of COVID-19 vaccine, and 8% were unvaccinated. During the 5–15 days after Paxlovid treatment was dispensed, six hospitalizations and 39 ED encounters considered to be related to SARS-CoV-2 infection were identified, representing <1% of all patients to whom Paxlovid treatment was dispensed during the study period. A higher proportion of those identified with COVID-19–related hospitalizations or ED encounters were either unvaccinated or vaccinated with 1 dose of COVID-19 vaccine (eight of 45; 17.8%) compared with all treated patients (514 of 5,287; 9.7%). Among the six hospitalized patients, five had received 3 doses of COVID-19 vaccine and one had received a single vaccine dose. All hospitalized patients had comorbidities or were of advanced age (range = 61–104 years), which put them at increased risk for severe COVID-19. Two hospitalized patients died; both were at high risk for severe illness because of multiple comorbidities and age, and their deaths were attributed to underlying disease. The remaining four hospitalized patients recovered, as did the 39 patients with COVID-19–related ED encounters during the 5–15 days after Paxlovid was dispensed. Third and fourth doses of mRNA COVID vaccines provided additional protection against moderate and severe COVID-19 during Omicron BA.1 and BA.2 subvariant surgesIn this MMWR, authors used data from the VISION network to review ED/urgent care visits and hospitalizations for COVID-19-like illness to evaluate the vaccine efficacy of 2, 3, and 4 doses of mRNA COVID-19 vaccines (Pfizer-BioNTech or Moderna) compared with no vaccination among adults without immunocompromising conditions. The data set included 214,487 emergency department/urgent care (ED/UC) visits and 58,782 hospitalizations with a COVID-19–like illness diagnosis among 10 states during December 18, 2021–June 10, 2022. VE against COVID-19–associated hospitalization 7–119 days and ≥120 days after receipt of dose 3 was 92% (95% CI = 91%–93%) and 85% (95% CI = 81%–89%), respectively, during the BA.1 period, compared with 69% (95% CI = 58%–76%) and 52% (95% CI = 44%–59%), respectively, during the BA.2/BA.2.12.1 period. Patterns were similar for ED/UC encounters. Among adults aged ≥50 years, VE against COVID-19–associated hospitalization ≥120 days after receipt of dose 3 was 55% (95% CI = 46%–62%) and ≥7 days (median = 27 days) after a fourth dose was 80% (95% CI = 71%–85%) during BA.2/BA.2.12.1 predominance. During the BA.1 period, VE against ED/urgent care visits declined to 73% ≥120 days (median = 132 days) after the third vaccine dose; during the BA.2/BA.12.1 period, VE declined to 26% at ≥120 days (median = 166 days) after the third dose. VE against hospitalization associated with a third mRNA vaccine dose was higher than that associated with a second vaccine dose but declined during both periods at ≥120 days to 85% during the BA.1 period (median = 132 days) and 52% during the BA.2/BA.2.12.1 period (median = 168 days). Among adults aged ≥50 years eligible to receive a fourth mRNA vaccine dose, VE for COVID-19–associated ED/UC encounters during the BA.2/BA.2.12.1 period was 32% at ≥120 days after the third dose (median interval = 170 days) but increased to 66% ≥7 days after the fourth dose (median interval = 28 days). VE against COVID-19–associated hospitalization was 55% ≥120 days after the third dose but increased to 80% ≥7 days after the fourth dose. COVID-19–associated hospitalization rates among disability-eligible Medicare beneficiaries were approximately 50% higher than rates among age-eligible (i.e., ≥65 years) beneficiariesIn this MMWR, researchers compared COVID-19 cases and hospitalizations during January 2020–November 2021, among beneficiaries aged ≥18 years who were either eligible because of a disability (disability-eligible) or only eligible because of age ≥65 years (age-eligible). COVID-19 incidence and hospitalization rates were higher in the disability-eligible group (10,978 and 3,148 per 100,000 population, respectively) throughout the study period compared with the age-eligible group (8,102 and 2,129 per 100,000 population, respectively). Both COVID-19 incidence and hospitalization rates increased with age in both disability- and age-eligible beneficiaries. American Indian or Alaska Native (AI/AN) persons had the highest disability-eligible (4,962 per 100,000) and age-eligible (5,024 per 100,000) hospitalization rates. Among all other racial and ethnic groups, hospitalization rates were higher among disability-eligible than among age-eligible patients. COVID-19 incidence and hospitalization rates among disability-eligible Medicare beneficiaries were disproportionally higher than rates among age-eligible beneficiaries. Overall, the in-hospital mortality rate was lower among disability-eligible patients (16.5%) than among age-eligible patients (19.0%). However, the mortality rate among disability-eligible beneficiaries aged ≥65 years was 19.1%, similar to that among age-eligible beneficiaries (19.0%). RECURRENT TOPICSProviding COVID-19 vaccinationsAll Alaskans and people who work or live in Alaska who are aged 6 months and older are eligible for vaccination against COVID-19. If you are interested in providing the COVID-19 vaccine in your office or clinic, please visit the COVID-19 Vaccination Program Provider Enrollment page. If you have additional questions, please email Matthew Bobo at matthew.bobo@alaska.gov Free self-paced online course available to promote COVID-19 vaccine confidence in Alaskan CommunitiesTwo online courses are available for free through the UAA Alaska Center for Rural Health & Health Workforce in partnership with the State of Alaska Department of Health and Social Services Division of Public Health. These courses are designed to provide participants the knowledge and skills to promote COVID-19 vaccine confidence among clients, family, friends, patients and/or community members in Alaskan communities. For those who complete the online course, there is an option to attend the live practical session to apply what they learned. These will occur monthly until the end of June and the live sessions are also free.
The Provider course is designed for licensed, or license eligible healthcare providers (1 contact hour, Interprofessional Continuing Education credit [ANMC]).
The Community course is aimed at entry level healthcare professionals and the interested public (3 hours) Oral COVID-19 AntiviralsThe FDA has authorized two oral antivirals – Paxlovid and Molnupiravir - for the treatment of mild-to-moderate COVID-19 in individuals who are at high risk for progression to severe COVID-19. Both treatments must be initiated within 5 days of symptom onset. Molnupiravir is authorized for adults (18 years and older) and Paxlovid is authorized adults and adolescents (12 years and older). Learn more at covidrx.alaska.gov. Long-Acting Monoclonal Antibodies for COVID-19 pre-exposure prophylaxis (PrEP)Evusheld (tixagevimab/cilgavimab) is a long-acting monoclonal antibody therapy that has been authorized by the FDA under EUA for pre-exposure prophylaxis for COVID-19. This product is given by intramuscular injection every six months and may help prevent COVID-19 infection. Evushelt is authorized for adults and adolescents (aged ≥12 years) who do not have SARS-CoV-2 infection, who have not been recently exposed to an individual with SARS-CoV-2 infection, AND who are moderately to severely immunocompromised or are not able to be fully vaccinated with any available COVID-19 vaccines due to a history of severe adverse reactions to a COVID-19 vaccine. For more information, read the NIH COVID-19 Treatment Guidelines Panel's statement on Evusheld. JAMA Patient Page on Evusheld PregnancyThe CDC strongly recommends COVID-19 vaccination either before or during pregnancy because the benefits of vaccination outweigh known or potential risks. For the latest recommendations, check out the CDC webpage on COVID in Pregnant and Recently Pregnant People and recommendations from the American College of Obstetrics and Gynecology. In addition, JAMA has published a one-page patient information sheet on COVID-19 and pregnancy. Post-acute Sequelae of COVID-19 (PASC)For the latest recommendations, check out the CDC webpage on Post-COVID-19 Syndrome and Evaluating and Caring for Patients with Post-COVID conditions. This page was most recently updated on May 5. COVID-19 Speakers’ BureauAnyone can request a free presentation for a group interested in learning more about the COVID-19 in Alaska. Aside from COVID-19Alaska Native Medical Center opens Alaska’s only burn centerUnder the leadership of Dr Brownson, ANMC has been added to bed counts and regional capabilities for the Western Region Burn Disaster Consortium (WRBDC). If there is a burn incident anticipated or occurring in which a surge of burn patients is possible please ensure your local burn center and whoever is responsible at the state level is aware prior to calling the 866 WRBDC number and requesting a partial or full activation. The WRBDC is currently in a stand-by/ready state as it is fire season and there are two Burn MDs on call in addition to the Coordinator team. Project FirstlineIf you or your organization are interested in learning more about infection control in health care, the CDC has a robust training collaborative and resource library under Project Firstline. If you’re interested in learning more about Project Firstline or potentially arranging a training session, please contact Kelley O’Leary at kelley.oleary@alaska.gov Provider Relief Fund – Request to report lateThe Provider Relief Fund (PRF) Request to Report Late Due to Extenuating Circumstances process is intended for providers who were required to report in an applicable reporting period, but extenuating circumstances prevented them from submitting a report by the required deadline. For each PRF Reporting Period, a provider may request an opportunity to complete their report after the reporting period deadline based on attestation that one of the following allowable extenuating circumstances applies at the time of the deadline: severe illness or death, impacted by natural disaster, lack of receipt of reporting communications, failure to click “submit,” internal miscommunication or error, or incomplete targeted distribution payments. Public Naloxone TrainingDHSS OSMAP Project HOPE are conducting public naloxone trainings as a community outreach and to increase awareness and access. Email projecthope@alaska.gov. Harm reduction strategies and resourcesThe Alaska Native Tribal Health Consortium (ANTHC) HIV/STD Prevention and Substance Misuse Prevention programs host free virtual gatherings to discuss harm reduction strategies and resources in Alaska. Guest speakers are Alaska-based experts in the field of harm reduction. Recordings of harm reduction trainings can be found here. Several useful materials specific to Alaska opioid safety are at the following webpage. Check it out and print out information to hang up in your community. Events/Conferences/PresentationsCDC COCA Call - Recommendations for the Novavax COVID-19 Vaccine Primary Series in Adults 18 and Older – July 28, 2-3pm ESTThe Novavax COVID-19 vaccine is the first COVID-19 protein subunit vaccine recommended for use in the United States that is built on a familiar type of vaccine technology. Other protein subunit vaccines have been used for more than 30 years in the United States, including for hepatitis B, HPV, and acellular pertussis (whooping cough) vaccines. During this COCA Call, presenters will discuss CDC’s new guidance on the Novavax COVID-19 vaccine for adults ages 18 years and older, including adults who are moderately or severely immunocompromised. Webinar Link: https://www.zoomgov.com/j/1612728470 Passcode: 663869 CDC COCA Call - Monkeypox Outbreak: Updates on the Epidemiology, Testing, Treatment, and Vaccination – July 26Presenters discussed updates on the epidemiology of the monkeypox outbreak, commercial testing capability, how to obtain and use TPOXX (tecovirimat) to treat monkeypox, and vaccine strategy. World Professional Association for Transgender Health Global Education Institute – Sexuality Workshop – July 29The WPATH GEI Online Sexuality Workshop will be led by Dr. Gail Knudson (University of British Columbia). This workshop will include: author highlights from Trans Sex: Clinical Approaches to Trans Sexualities and Erotic Embodiments (Routledge, 2021); working with TGD children, adolescents, adults, and older adults, and specific generational concerns; ethical principles involved in Sexual Health Service delivery; how the PLISSIT MODEL and ISSWSH Process of Care are applicable to TGD clients in general practice; SOC 8 Sexual Health Chapter Recommendations; possible effects of the hormone therapy and other medications on sexual pleasure; overview of international associations’ position statements and declarations supporting sexuality and sexual health of TGD persons, as well as case studies. Medical and Mental Health CEs are available for this GEI Workshop.
Attendees must be available to attend the live session "in-person" via Zoom on Friday, July 29, 2022 from 11:00am - 3:00pm EDT, in order to complete the WPATH GEI Online Sexuality Workshop. Registration Link Drug Addiction Treatment Act (DATA) Waivers ECHOUAA Center for Human Development and the State of Alaska Division of Behavioral Health are pleased to offer a new Project ECHO for medical providers who are currently DATA waivered and/or medical providers who are interested in obtaining a DATA waiver. August 10 at 12:00pm Prenatal/OB MAT Sept 14 at 12:00pm Understanding and reducing buprenorphine diversion Registration link for DATA Waivers ECHO: https://echo.zoom.us/meeting/register/tJYqce-gqDovGdDitNu5EPxMU1UOPrBth7Wv
Hosted and facilitated by UAA’S Center for Human Development Alaska ECHO Project in partnership with the State of Alaska Division of Behavioral Health. For questions, please contact echo@alaskachd.org ANTHC Tribal Health Webinar SeriesThe ANTHC Tribal Health Webinar series occurs on Friday from 12-1pm on Zoom and is open to the public. There are no webinars throughout the remainder of June or in the month of July since many individuals will be participating in subsistence activities during these months. The series will resume in August. CDC Clinical Support: There is a Clinician On-Call Center, a 24-hour hotline with trained CDC clinicians standing by to answer COVID-19 questions. Call 1-800-CDC-INFO (800-232-4636) and ask for the Clinician On-Call Center. The most up-to-date, evidence based COVID-19 treatment guidelines can be found at:
NIH COVID-19 Treatment Guidelines
IDSA Guidelines on the Treatment and Management of Patient with COVID-19 Alaska Responders Relief Line (844) 985-8275
Your well-being matters. Your behavioral health colleagues are standing by to talk 24/7: 844-985-8275
Recognizing the unique stressors that providers face, the Division of Behavioral Health has established a 24/7 support line, (844) 985-8275, for healthcare and behavioral professionals impacted by COVID personally and professionally. Staff supporting the call line recognize callers are often first responders and will allow callers to openly express their experiences and feelings serving Alaskans impacted by COVID. This service is also available to immediate family members of first responders who may be experiencing stress, anxiety and other hard to label emotions as a result of their loved one engaging on the front lines. |



